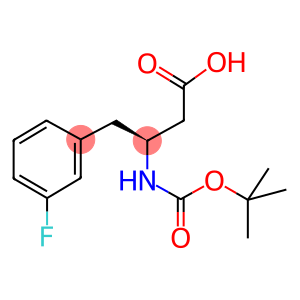
N-T-BUTOXYCARBONYL-(S)-3-AMINO-4-(3-FLUOROPHENYL)BUTANOIC ACID
Boc-(S)-3-Amino-4-(3-fluoro-phenyl)-butyric acid
CAS: 270596-51-5
Molecular Formula: C15H20FNO4
N-T-BUTOXYCARBONYL-(S)-3-AMINO-4-(3-FLUOROPHENYL)BUTANOIC ACID - Names and Identifiers
N-T-BUTOXYCARBONYL-(S)-3-AMINO-4-(3-FLUOROPHENYL)BUTANOIC ACID - Physico-chemical Properties
| Molecular Formula | C15H20FNO4 |
| Molar Mass | 297.32 |
| Storage Condition | 2-8°C |
N-T-BUTOXYCARBONYL-(S)-3-AMINO-4-(3-FLUOROPHENYL)BUTANOIC ACID - Risk and Safety
| Hazard Class | IRRITANT |
N-T-BUTOXYCARBONYL-(S)-3-AMINO-4-(3-FLUOROPHENYL)BUTANOIC ACID - Introduction
Boc-(S)-3-Amino-4-(3-fluoro-phenyl)-butyric acid is an organic compound with the following properties:
1. Appearance: colorless solid.
2. molecular formula: C. H. FNO.
3. Molecular weight: 313.33g/mol.
4. Solubility: Soluble in some polar organic solvents, such as dimethylformamide (DMF) and dichloromethane.
5. Melting point: between about 92-94°C.
Boc-(S)-3-Amino-4-(3-fluoro-phenyl)-butyric acid is widely used in organic synthesis and medicinal chemistry. Its main uses include:
1. drug intermediates: as intermediates in drug synthesis, used for the synthesis of biologically active compounds, such as anticancer drugs, hormone drugs.
2. peptide synthesis: as an amino acid protecting group, used in peptide synthesis reaction, functional groups on amino acid side chains, such as carboxyl and amino groups, can be protected to prevent non-specific reactions.
the common method for preparing Boc-(S)-3-Amino-4-(3-fluoro-phenyl)-butyric acid is to first react amino-acrylate and 3-fluorobenzaldehyde to obtain the corresponding alcohol, and then react with Boc-ethyl imidate in the presence of sodium bicarbonate to obtain the target product.
For safety information, the specific safety evaluation of Boc-(S)-3-Amino-4-(3-fluoro-phenyl)-butyric acid may require further experimental data and expertise. In use, need to pay attention to the following matters:
1. storage: should be stored in a dry, cool place, avoid direct sunlight.
2. operation: operation should wear appropriate personal protective equipment, such as gloves, protective glasses, etc.
3. waste disposal: in accordance with local environmental regulations, properly dispose of waste to avoid environmental pollution.
When using and handling this compound, it is recommended to follow laboratory safety procedures and consult a professional.
1. Appearance: colorless solid.
2. molecular formula: C. H. FNO.
3. Molecular weight: 313.33g/mol.
4. Solubility: Soluble in some polar organic solvents, such as dimethylformamide (DMF) and dichloromethane.
5. Melting point: between about 92-94°C.
Boc-(S)-3-Amino-4-(3-fluoro-phenyl)-butyric acid is widely used in organic synthesis and medicinal chemistry. Its main uses include:
1. drug intermediates: as intermediates in drug synthesis, used for the synthesis of biologically active compounds, such as anticancer drugs, hormone drugs.
2. peptide synthesis: as an amino acid protecting group, used in peptide synthesis reaction, functional groups on amino acid side chains, such as carboxyl and amino groups, can be protected to prevent non-specific reactions.
the common method for preparing Boc-(S)-3-Amino-4-(3-fluoro-phenyl)-butyric acid is to first react amino-acrylate and 3-fluorobenzaldehyde to obtain the corresponding alcohol, and then react with Boc-ethyl imidate in the presence of sodium bicarbonate to obtain the target product.
For safety information, the specific safety evaluation of Boc-(S)-3-Amino-4-(3-fluoro-phenyl)-butyric acid may require further experimental data and expertise. In use, need to pay attention to the following matters:
1. storage: should be stored in a dry, cool place, avoid direct sunlight.
2. operation: operation should wear appropriate personal protective equipment, such as gloves, protective glasses, etc.
3. waste disposal: in accordance with local environmental regulations, properly dispose of waste to avoid environmental pollution.
When using and handling this compound, it is recommended to follow laboratory safety procedures and consult a professional.
Last Update:2024-04-09 18:58:34
Supplier List
Spot supply
Product Name: Boc-(S)-3-Amino-4-(3-fluoro-phenyl)-butyric acid Visit Supplier Webpage Request for quotationCAS: 270596-51-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Boc-(S)-3-Amino-4-(3-fluoro-phenyl)-butyric acid Request for quotation
CAS: 270596-51-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 270596-51-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: BOC-(S)-3-AMINO-4-(3-FLUOROPHENYL)BUTYRIC ACID Request for quotation
CAS: 270596-51-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 270596-51-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: BOC-(S)-3-AMINO-4-(3-FLUOROPHENYL)BUTYRIC ACID Visit Supplier Webpage Request for quotation
CAS: 270596-51-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 270596-51-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Boc-(S)-3-Amino-4-(3-fluoro-phenyl)-butyric acid Visit Supplier Webpage Request for quotationCAS: 270596-51-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Boc-(S)-3-Amino-4-(3-fluoro-phenyl)-butyric acid Request for quotation
CAS: 270596-51-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 270596-51-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: BOC-(S)-3-AMINO-4-(3-FLUOROPHENYL)BUTYRIC ACID Request for quotation
CAS: 270596-51-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 270596-51-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: BOC-(S)-3-AMINO-4-(3-FLUOROPHENYL)BUTYRIC ACID Visit Supplier Webpage Request for quotation
CAS: 270596-51-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 270596-51-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
N-T-BUTOXYCARBONYL-(S)-3-AMINO-4-(3-FLUOROPHENYL)BUTANOIC ACID
CS-2249
N-Methylaminobarbital
1-(乙氧羰基)环丙烷甲酸
2,7-Diazaspiro[4.4]nonane,dihydrobroMide
1,3,5-Triazine-2,4-diamine, 6-chloro-N2,N2-dipropyl-
咪唑啉季铵化物ODD
Ageratone
1,3,4,6-Tetrakis(methoxymethyl)glycoluril
蚁酸铵
CS-2249
N-Methylaminobarbital
1-(乙氧羰基)环丙烷甲酸
2,7-Diazaspiro[4.4]nonane,dihydrobroMide
1,3,5-Triazine-2,4-diamine, 6-chloro-N2,N2-dipropyl-
咪唑啉季铵化物ODD
Ageratone
1,3,4,6-Tetrakis(methoxymethyl)glycoluril
蚁酸铵



